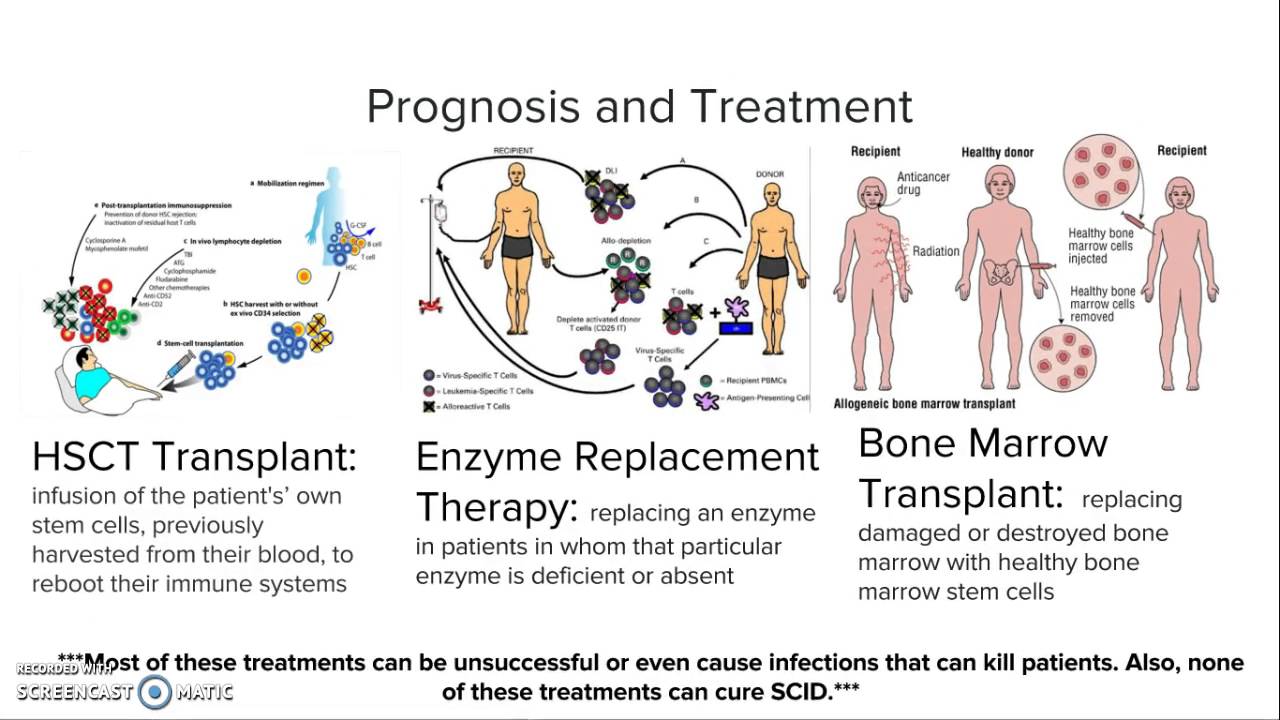
Once discovered, a child with SCID is usually should be kept in protective isolation to prevent catching a possibly fatal viral infection.Īt this time the only known effective and curative therapy is hematopoietic stem cell transplantation (HSCT) from a healthy donor. The most severe is severe combined immune deficiency (SCID), where both the disease-fighting immune system T-cells and B-cells are absent or broken. What is severe combined immune deficiency (SCID)?īecause the human immune system is so complex, there are many possible genetic mutations that can result in severe congenital immune deficiency. When present, SCID and other congenital severe immune deficiencies, like Wiskott Aldrich Syndrome, are life-threatening diseases because the child cannot effectively fight off common infections.Ĭhildren with severe immune deficiencies often need to be hospitalized and can die from common respiratory viruses like flu, RSV or adenovirus, other common viruses like chickenpox or opportunistic infections that only affect the severely immune-compromised (people with weak immune systems) like certain types of pneumonia. This condition is called an immune deficiency or severe combined immune deficiencies (SCID). 2006 8(5) Suppl: S12-S252 as authored by the American College of Medical Genetics (ACMG) and commissioned by the Health Resources and Services Administration (HRSA).These are a group of genetic diseases in which a child is born without a normal ability to fight infection. 
Selection of conditions based upon Newborn Screening: Towards a Uniform Screening Panel and System.Newborn blood spot screening for severe combined immunodeficiency by measurement of T-cell receptor excision circles Approved guideline. Clinical and Laboratory Standards Institute (CLSI).laboratories currently screening for SCID. The Division of Laboratory Sciences continues to conduct proficiency testing programs with quarterly send outs to all U.S. Increase number of babies screened for SCIDĬDC funding has helped states screen for more than one million babies for SCID.Increase number of laboratories screening for SCID.SCID newborn screening program activities continue to: The primary goal of CDC’s newborn screening program is to improve the health and lives of newborns. In 2015, CDC funded New York to develop advanced laboratory screening techniques and quality assurance materials that will improve the detection of SCID. Provided evidence to support the addition of SCID to the RUSP.Served as models for other states to implement population-based screening, and.Ensured SCID screening tests were ready for nationwide use,.Successful newborn screening pilot studies in these states have: Louisiana, Nevada, and North Carolina (2015).Florida, Illinois, New Jersey, North Carolina, Texas (2017).Eligible state or territorial programs are those that have not previously conducted state-wide SCID newborn screening and demonstrate sufficient laboratory expertise, facilities, and legal authority to conduct screening. Sponsors conferences and meetings to educate scientist about methods and reference materials for SCID testing.ĬDC’s Division of Laboratory Sciences funds SCID screening by state public health laboratories through cooperative agreements with newborn screening programs.Provides testing materials to labs to help ensure accurate SCID screening, and.Develops new laboratory methods to identify babies with SCID,.Provides training and technical assistance to state labs screening for SCID,.Funds SCID test development and early screening in collaboration with states,.In an effort to expand and improve screening, CDC’s Division of Laboratory Sciences: 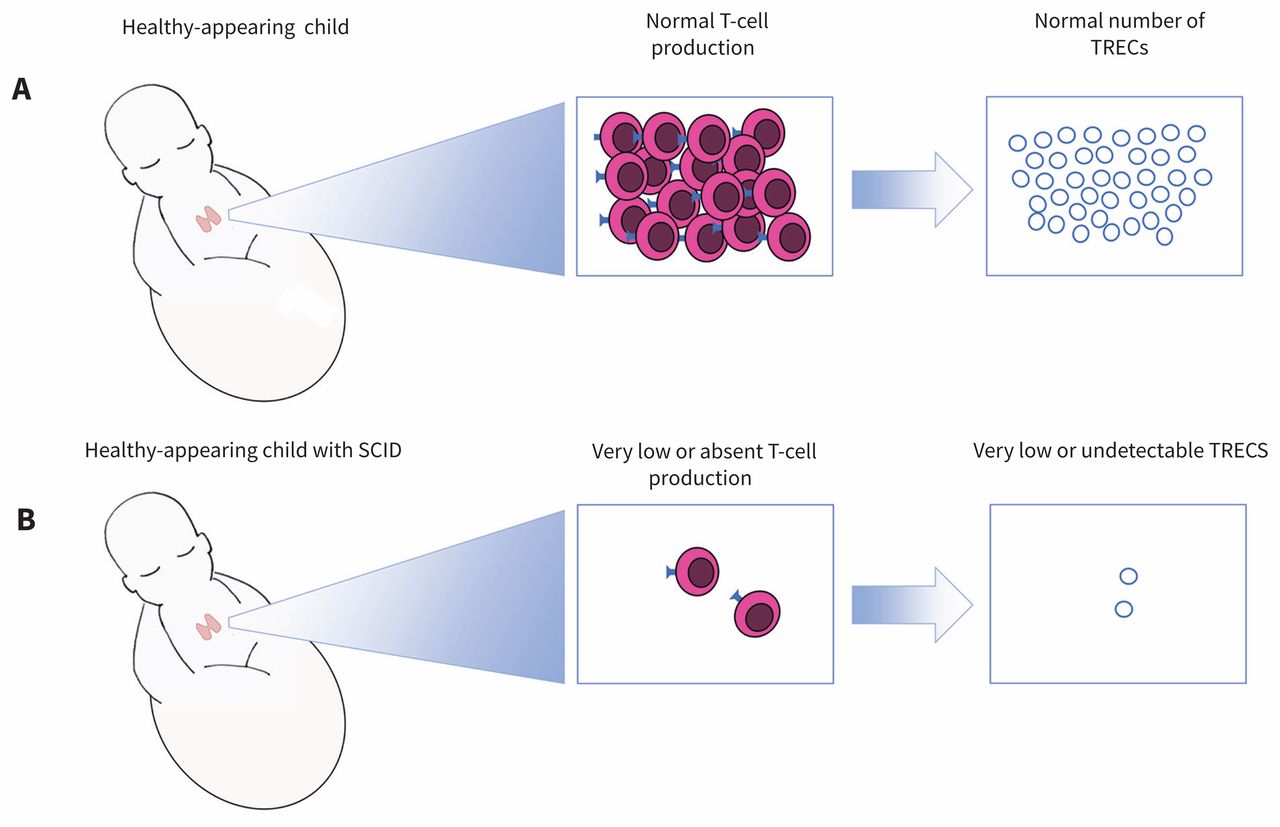
Since 2008, CDC has worked to increase the number of U.S states and territories that include accurate SCID testing as part of their newborn screening programs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
