|
The mass of water is almost equal to its volume at room temperature and the water remains in. at the temperature of 20 C to 25 C the density of the water is 0.9982 g/cc, or 998.2 kg/m 3. Actually, the exact density of water is not really 1 g/ml, but rather a bit less (very, very little less), at 0.9998395 g/ml at 4.0 Celsius (39.2 Fahrenheit). It is concluded that the density of water is a constant value, that is, 1 g/ml or 1 g/cm 3. Density is determined by dividing the mass of a substance by its volume:ĭensity is commonly expressed in units of g/cm 3 for solids, g/mL for liquids, and g/L for gases. A common unit of measurement for water's density is gram per milliliter (1 g/ml) or 1 gram per cubic centimeter (1 g/cm 3). Jobs Density of water is the ratio of the mass and volume of the water at room temperature is 998.2 kg/m 3. For example, the density of a gold coin and a gold statue are the same, even though the gold statue consists of the greater quantity of gold. An intensive property is one that is independent of the amount of matter present. Since pure substances have unique density values, measuring the density of a substance can help identify that substance. Physical properties can be measured without changing the chemical identity of the substance. Density is a physical property of matter. When water is used as the reference substance, the specific gravity of a substance determines whether it will sink or float. Where the volume water displaced is equal to the volume of solid.ĭensity is defined as the mass per unit volume of a substance. At 4 degrees Celsius, the density of water is 1 g/cm 3. The volume water displaced is equal to the difference between the final volume and the initial volume, or: Measuring the Volume of an Irregularly Shaped Solid Note that when measuring liquid volumes, it is important to read the graduated scale from the lowest point of the curved surface of the liquid, known as the liquid meniscus. In this lab, a beaker, two graduated cylinders and a burette will be used to measure liquid volumes, and their precision will be compared. The density of water (H 2 O) changes depending on temperature and pressure - as with other substances. The volume of a liquid can be directly measured with specialized glassware, typically in units of milliliters (mL) or liters (L). Data chart with the density of water at different temperatures. An extensive property is one that is dependent on the amount of matter present.  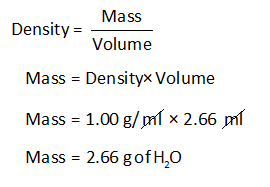 
Volume is the amount of space occupied by matter. In this lab, students will also determine the density of water as well as aluminum.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
